| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| 靶点 |
mitochondria-targeted superoxide dismutase mimetic
|
|---|---|
| 体外研究 (In Vitro) |
Mito-tempo(MT)是一种线粒体靶向的超氧化物歧化酶模拟物,通过抑制过氧亚硝酸盐的形成来保护对乙酰氨基酚(APAP)肝毒性的早期阶段。
|
| 体内研究 (In Vivo) |
在这两个时间点,Mito-TEMPO (MT) 均显着抑制 ALT 活性的上升并减少坏死区域,表明 Mito-TEMPO 的保护作用在 APAP 后持续至少 24 小时。在 APAP 肝毒性的后期,Mito-Tempo 可引起继发性细胞凋亡。通过阻断 RIP3,Mito-Tempo 会导致二次细胞凋亡,以响应过多的 APAP[1]。
|
| 酶活实验 |
半胱氨酸蛋白酶活性测定和蛋白质印迹
如所述测量肝脏半胱天冬酶活性(Lawson等人,1999)。简言之,将冷冻肝组织在含有5mM EDTA、2mM DTT和0.1%CHAPS的25mM HEPES缓冲液中匀浆,然后离心得到匀浆。将荧光底物(Ac-DEVD-AFC)加入到匀浆中,并在存在或不存在泛胱天蛋白酶抑制剂(z-VAD-fmk)的情况下测量荧光。结果表示为每单位时间每毫克蛋白质浓度的RFU。使用兔抗胱天蛋白酶3抗体和兔抗β-肌动蛋白抗体以及抗RIP3抗体,如所述(Bajt等,2000)进行蛋白质印迹。使用山羊抗兔HRP缀合的抗体对蛋白质进行可视化。
|
| 细胞实验 |
组织学[1]
将石蜡包埋的肝组织样本切成5μm切片,用苏木精和伊红(H&E)染色,以评估凋亡与坏死(Gujral等人,2002)。如前所述(Knight等人,2002),使用兔多克隆抗硝基酪氨酸抗体和Dako LSAB过氧化物酶试剂盒进行硝基酪氨酸染色。使用切割的胱天蛋白酶-3(Asp175)抗体进行活性胱天蛋白酶3染色。按照制造商的说明,使用原位细胞死亡检测试剂盒AP对细胞死亡进行末端脱氧核苷酸转移酶dUTP缺口末端标记(TUNEL)染色。
|
| 动物实验 |
Animals
Male C57BL/6J mice 8-12 weeks of age were kept in an environmentally controlled room with a 12h light/dark cycle. RIP3-deficient mice (C57BL/6N background) and C57BL/6N wild type animals were were acclimated before experiments with free access to diet and water. Experimental design Overnight fasted mice (16-18h) were treated i.p. with 300 mg/kg APAP dissolved in warm saline. Some mice were treated with 200mg/kg APAP in experiments evaluating effect of RIP3 deficiency. A dose of 20 mg/kg Mito-Tempo dissolved in saline was administered i.p. 1.5 or 3 h after APAP. Some mice were subsequently treated (i.p.) with 10 mg/kg Z-VD fmk (EP1013) dissolved in Tris-buffered saline or vehicle 2 h after APAP. To mimic the clinical care of APAP-overdose patients, some mice received the antidote NAC (i.p., 500 mg/kg) at 1.5 or 3 h after APAP overdose. Groups of mice were euthanized at 0-24 h post-APAP by exsanguination under isoflurane anesthesia. Additional mice were treated i.p. with 100 μg/kg Salmonella abortus equi endotoxin (ET) and 700 mg/kg galactosamine (Gal) for 6 h. Blood was drawn into a heparinized syringe and centrifuged to obtain plasma. Plasma ALT activities were measured using the ALT assay kit from Pointe Scientific, MI. The liver tissue was cut into pieces and fixed in 10% phosphate-buffered formalin for histology or flash frozen in liquid nitrogen and subsequently stored at −80°C. In vivo morpholino treatment The antisense sequence used for RIP3 was 5’-TAGGCCATAACTTGACAGAAGACAT-3’. The standard control in vivo oligo sequence from Gene Tools was used for all control morpholino treatments. Morpholinos were used as provided by the manufacturer and administered ip to mice (12.5 mg/kg body weight) every 24h for 2 days. Treatment with APAP was then done on day 3. |
| 参考文献 |
|
| 其他信息 |
Researchers previously reported that delayed treatment with Mito-tempo (MT), a mitochondria-targeted superoxide dismutase mimetic, protects against the early phase of acetaminophen (APAP) hepatotoxicity by inhibiting peroxynitrite formation. However, whether this protection is sustained to the late phase of toxicity is unknown. To investigate the late protection, C57Bl/6J mice were treated with 300 mg/kg APAP followed by 20 mg/kg MT 1.5 h or 3 h later. We found that both MT treatments protected against the late phase of APAP hepatotoxicity at 12 and 24 h. Surprisingly, MT-treated mice demonstrated a significant increase in apoptotic hepatocytes, while the necrotic phenotype was observed almost exclusively in mice treated with APAP alone. In addition, there was a significant increase in caspase-3 activity and cleavage in the livers of MT-treated mice. Immunostaining for active caspase-3 revealed that the positively stained hepatocytes were exclusively in centrilobular areas. Treatment with the pan-caspase inhibitor ZVD-fmk (10 mg/kg) 2 h post-APAP neutralized this caspase activation and provided additional protection against APAP hepatotoxicity. Treatment with N-acetylcysteine, the current standard of care for APAP poisoning, protected but did not induce this apoptotic phenotype. Mechanistically, MT treatment inhibited APAP-induced RIP3 kinase expression, and RIP3-deficient mice showed caspase activation and apoptotic morphology in hepatocytes analogous to MT treatment. These data suggest that while necrosis is the primary cause of cell death after APAP hepatotoxicity, treatment with the antioxidant MT may switch the mode of cell death to secondary apoptosis in some cells. Modulation of mitochondrial oxidative stress and RIP3 kinase expression play critical roles in this switch.[1]
Mitochondrial oxidative damage contributes to a range of degenerative diseases. Consequently, the selective inhibition of mitochondrial oxidative damage is a promising therapeutic strategy. One way to do this is to invent antioxidants that are selectively accumulated into mitochondria within patients. Such mitochondria-targeted antioxidants have been developed by conjugating the lipophilic triphenylphosphonium cation to an antioxidant moiety, such as ubiquinol or alpha-tocopherol. These compounds pass easily through all biological membranes, including the blood-brain barrier, and into muscle cells and thus reach those tissues most affected by mitochondrial oxidative damage. Furthermore, because of their positive charge they are accumulated several-hundredfold within mitochondria driven by the membrane potential, enhancing the protection of mitochondria from oxidative damage. These compounds protect mitochondria from damage following oral delivery and may therefore form the basis for mitochondria-protective therapies. Here we review the background and work to date on this class of mitochondria-targeted antioxidants.[2] |
| 分子式 |
C29H37CLN2O3P
|
|---|---|
| 分子量 |
528.04
|
| 精确质量 |
528.23
|
| CAS号 |
1569257-94-8
|
| 相关CAS号 |
Mito-TEMPO;1334850-99-5
|
| PubChem CID |
73504624
|
| 外观&性状 |
Orange to red solid powder
|
| tPSA |
34.3
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
6
|
| 重原子数目 |
36
|
| 分子复杂度/Complexity |
612
|
| 定义原子立体中心数目 |
0
|
| SMILES |
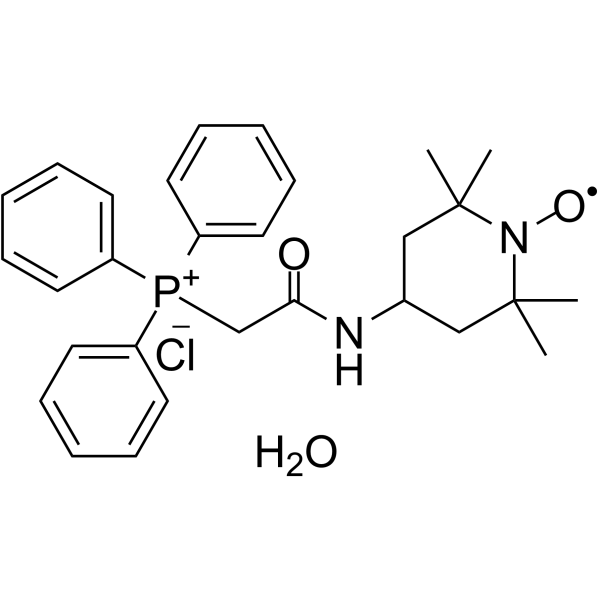
C([P+](C1C=CC=CC=1)(C1C=CC=CC=1)C1C=CC=CC=1)C(NC1CC(C)(C)N([O])C(C)(C)C1)=O.[Cl-].O |^1:28|
|
| InChi Key |
AIAJCDXYZBOVGT-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C29H34N2O2P.ClH.H2O/c1-28(2)20-23(21-29(3,4)31(28)33)30-27(32)22-34(24-14-8-5-9-15-24,25-16-10-6-11-17-25)26-18-12-7-13-19-26;;/h5-19,23H,20-22H2,1-4H3;1H;1H2
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8938 mL | 9.4690 mL | 18.9380 mL | |
| 5 mM | 0.3788 mL | 1.8938 mL | 3.7876 mL | |
| 10 mM | 0.1894 mL | 0.9469 mL | 1.8938 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。