| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| 靶点 |
Na+-K+-Cl+ cotransporter (NKCC); hNKCC1A (IC50 = 0.68 μM); hNKCC2A (IC50 = 4.0 μM)
|
|---|---|
| 体外研究 (In Vitro) |
NKCC 的两种主要人类剪接变体 hNKCC1A 和 hNKCC2A 均受到布美他尼钠的抑制 [1]。在表达 NKCC1A 的卵母细胞中,布美他尼钠(0.03-100 μM;5 分钟)以剂量依赖性方式减少 86Rb+ 的摄取 [1]。在 HEK-293 细胞中,布美他尼钠抑制 NKCC2 同工型 B,IC50 值为 0.54 μM[2]。
Na(+)- k (+)-Cl(-)共转运体NKCC1在调节神经元内Cl(-)浓度中起主要作用。NKCC1功能异常与包括癫痫在内的几种脑部疾病有关。布美他尼是唯一可用的选择性NKCC1抑制剂,但也抑制NKCC2,在治疗脑部疾病时可引起严重的不良反应。因此,选择nkcc1的布美他胺衍生物将是一个理想的选择。在本研究中,我们利用非洲爪蟾卵母细胞异种表达系统,比较了布美他尼及其衍生物对NKCCs两种主要人类剪接变体hNKCC1A和hNKCC2A的影响。这些衍生物是从约5000个3-氨基-5-磺胺基苯甲酸衍生物中挑选出来的,涵盖了广泛的结构修饰和利尿作用。据我们所知,这种结构-功能关系之前还没有在NKCC1中进行过。布美他尼对hNKCC1A的半数最大抑制浓度(ic50)分别为0.68 μ m (hNKCC1A)和4.0μM (hNKCC2A),表明该药对hNKCC1A的抑制效力是hNKCC2A的6倍。布美他胺分子的侧链取代对抑制hNKCC1A的效力有不同程度的影响。这允许定义配体相互作用所需的最小结构要求。出乎意料的是,只有少数布美他胺衍生物比布美他胺更有效地抑制hNKCC1A,而且大多数也抑制hNKCC2A,两种NKCC亚型的ic50之间存在高度显著的相关性。这些数据表明,抑制NKCC1和NKCC2的结构要求相似,这使得开发对NKCC1具有高选择性的布美他尼相关化合物变得复杂。[1] |
| 体内研究 (In Vivo) |
布美他尼钠(7.6-30.4 mg/kg;静脉注射)减轻了皮质和纹状体表观扩散系数 (ADC) 比率的下降(减少 40-67%),表明水肿形成减少[3]。布美他尼钠还可以减少梗塞面积[3]。布美他尼钠在大鼠静脉注射2 mg/kg、8 mg/kg和20 mg/kg后表现出不同的半衰期,分别为21.4分钟、53.8分钟和137分钟[4]。
|
| 酶活实验 |
静脉注射布美他尼(7.6-30.4 mg/kg)可防止皮肤和纹状体表观扩散系数(ADC)降低40-67%,这是水肿发展减少的标志[3]。此外,布美他尼的静脉剂量可以减少至 2 mg/kg、8 mg/kg 和 20 mg/kg [4]。
闭塞前立即静脉注射布美他尼(7.6-30.4 mg/kg)可减弱皮质和纹状体ADC比率的下降(40-67%),表明水肿形成减少。通过TTC染色,布美他尼还可减小梗死面积。这些发现表明,脑缺血时,管腔血脑屏障Na-K-Cl共转运蛋白参与了水肿的形成。[3] 布美他尼2、8、20 mg/kg静脉和口服,测定布美他尼在大鼠体内的药代动力学和药效学(n = 10-12)。研究了布美他尼在胃肠道各部位的吸收情况,以及口服布美他尼后血药浓度出现多峰的原因。静脉给药后,布美他尼的药代动力学参数t1/2(21.4、53.8 vs. 127 min)、CL(35.8、19.1 vs. 13.4 ml/min /kg)、CLNR(35.2、17.8 vs. 12.6 ml/min /kg)和VSS (392,250 vs. 274 ml/kg)在研究剂量范围内呈剂量依赖性。这可能与布美他胺在大鼠体内的饱和代谢有关。在静脉给药后,每100克体重的8小时尿量随着剂量的增加而显著增加,这可能是由于随着剂量的增加,8小时尿中布美他胺的排泄量显著增加。给药8 mg/kg后,每100 g体重8小时尿液中钠和氯的排泄总量也显著增加,而钾的排泄值则与剂量无关。口服给药后,布美他尼在24小时尿液中排泄的剂量百分比与剂量无关。布美他尼从胃肠道的所有区域吸收,分别在口服剂量为2、8和20 mg/kg后的1至24小时内,约有43.7%、50.0%和38.4%的口服剂量被吸收。因此,口服给药后出现多峰可能主要与胃排空模式有关。[4] |
| 细胞实验 |
NKCC1A活性测定[1]
为了在摄取实验之前激活NKCC1A,将表达hnkcc1a的卵母细胞或未注射的对照卵母细胞(每孔5 - 15个卵母细胞)在室温下在高渗透性无K+溶液中预孵育30分钟(mM: 5氯化胆碱,95 NaCl, 1 MgCl2, 1 CaCl2, 10 Hepes;pH 7.4, 207 mOsm),导致卵母细胞收缩,从而激活NKCC1A。为了测量K+内流,将卵母细胞暴露于等渗测试溶液中,其中KCl (5 mM)取代氯化胆碱,并加入2-3 μCi/mL 86Rb+作为K+的示踪剂。用15型自动渗透计对测试介质的渗透性进行了验证。在测试溶液中加入Bumetanide/布美他尼(0.03 ~ 100 μM)、其衍生物(1 ~ 100 μM)或对照物(≤1%,确保实验中所有被测卵母细胞均暴露于相关药物溶剂中)。摄取实验在室温下进行,轻微搅拌5分钟,我们已经证明这是在K+摄取的线性阶段。在不含86Rb+的冷冻实验液中快速洗涤3次,终止内流实验,然后将卵母细胞单独溶解于200 μL 10%十二烷基硫酸钠中,置于闪烁小瓶中。采用Tri-Carb 2900TR液体闪烁分析仪,用Ultima Gold XR闪烁液进行液体闪烁β计数。人类NKCC1剪接变异体a介导的K+摄取被评估为([在x μM药物存在下表达fluxnkcc1的卵母细胞]-[在x μM药物存在下注射fluxnkcc1的卵母细胞]),以纠正内源性NKCC活性。所有实验至少重复3次(范围:3-6次)。 |
| 动物实验 |
Animal/Disease Models: Normotensive SD (SD (Sprague-Dawley)) rats (250-300 g) [3]
Doses: 7.6 mg/kg, 15.2 mg/kg, 30.4 mg/kg Route of Administration: intravenous (iv) (iv)injection Experimental Results: diminished middle cerebral artery occlusion (MCAO) ) caused a decrease in ADC values in all four ipsilateral regions (L1-L4). Animal/Disease Models: Male SD (SD (Sprague-Dawley)) rat (220-300 g) [4] Doses: 2 mg/kg, 8 mg/kg, 20 mg/kg (pharmacokinetic/PK/PK analysis) Route of Administration: intravenous (iv) (iv)administration Experimental Results: T1/2 (21.4 minutes, 53.8 minutes and 137 minutes for 2 mg/kg, 8 mg/kg and 20 mg/kg respectively) |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Bumetanide is completely absorbed (80%), and the absorption is not altered when taken with food. Bioavailability is almost complete. Oral administration of carbon-14 labeled Bumex to human volunteers revealed that 81% of the administered radioactivity was excreted in the urine, 45% of it as unchanged drug. Biliary excretion of Bumex amounted to only 2% of the administered dose. 0.2 - 1.1 mL/min/kg [preterm and full-term neonates with respiratory disorders] 2.17 mL/min/kg [neonates receiving bumetanide for volume overload] 1.8 +/- 0.3 mL/min/kg [geriatric subjects] 2.9 +/- 0.2 mL/min/kg [younger subjects] Metabolism / Metabolites 45% is secreted unchanged. Urinary and biliary metabolites are formed by oxidation of the N-butyl side chain. Biological Half-Life 60-90 minutes |
| 毒性/毒理 (Toxicokinetics/TK) |
Effects During Pregnancy and Lactation
◉ Summary of Use during Lactation It is unknown if bumetanide is excreted into breastmilk. It should be avoided while breastfeeding a newborn because it may decrease milk flow or completely suppress lactation. Low doses in mothers whose lactation is well established are unlikely to suppress lactation. In general, alternate drugs are preferred. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information on bumetanide was not found as of the revision date. Intense diuresis, fluid restriction and breast binding have been used to suppress lactation immediately postpartum. The added contribution of the diuretic to the other measures, which are effective in suppressing lactation, has not been studied. No data exist on the effects of loop diuretics on established, ongoing lactation. Protein Binding 97% |
| 参考文献 |
|
| 其他信息 |
Bumetanide is a member of the class of benzoic acids that is 4-phenoxybenzoic acid in which the hydrogens ortho to the phenoxy group are substituted by butylamino and sulfamoyl groups. Bumetanide is a diuretic, and is used for treatment of oedema associated with congestive heart failure, hepatic and renal disease. It has a role as a diuretic and an EC 3.6.3.49 (channel-conductance-controlling ATPase) inhibitor. It is a sulfonamide, an amino acid and a member of benzoic acids.
Bumetanide is a sulfamyl diuretic. Bumetanide is a Loop Diuretic. The physiologic effect of bumetanide is by means of Increased Diuresis at Loop of Henle. Bumetanide is a potent sulfamoylanthranilic acid derivative belonging to the class of loop diuretics. In the brain, bumetanide may prevent seizures in neonates by blocking the bumetanide-sensitive sodium-potassium-chloride cotransporter (NKCC1), thereby inhibiting chloride uptake thus, decreasing the internal chloride concentration in neurons and may block the excitatory effect of GABA in neonates. A sulfamyl diuretic. Drug Indication For the treatment of edema associated with congestive heart failure, hepatic and renal disease including the nephrotic syndrome. FDA Label Treatment of autism spectrum disorder Mechanism of Action Bumetanide interferes with renal cAMP and/or inhibits the sodium-potassium ATPase pump. Bumetanide appears to block the active reabsorption of chloride and possibly sodium in the ascending loop of Henle, altering electrolyte transfer in the proximal tubule. This results in excretion of sodium, chloride, and water and, hence, diuresis. Pharmacodynamics Bumetanide is a loop diuretic of the sulfamyl category to treat heart failure. It is often used in patients in whom high doses of furosemide are ineffective. There is however no reason not to use bumetanide as a first choice drug. The main difference between the two substances is in bioavailability. Bumetanide has more predictable pharmacokinetic properties as well as clinical effect. In patients with normal renal function, bumetanide is 40 times more effective than furosemide. Increased transport of Na+ across an intact blood-brain barrier (BBB) participates in edema formation during the early hours of cerebral ischemia. In previous studies, the authors showed that the BBB Na-K-Cl cotransporter is stimulated by factors present during ischemia, suggesting that the cotransporter may contribute to the increased brain Na+ uptake in edema. The present study was conducted to determine (1) whether the Na-K-Cl cotransporter is located in the luminal membrane of the BBB, and (2) whether inhibition of the BBB cotransporter reduces brain edema formation. Perfusion-fixed rat brains were examined for cotransporter distribution by immunoelectron microscopy. Cerebral edema was evaluated in rats subjected to permanent middle cerebral artery occlusion (MCAO) by magnetic resonance diffusion-weighted imaging and calculation of apparent diffusion coefficients (ADC). The immunoelectron microscopy studies revealed a predominant (80%) luminal membrane distribution of the cotransporter. Magnetic resonance imaging studies showed ADC ratios (ipsilateral MCAO/contralateral control) ranging from 0.577 to 0.637 in cortex and striatum, indicating substantial edema formation. Intravenous bumetanide (7.6-30.4 mg/kg) given immediately before occlusion attenuated the decrease in ADC ratios for both cortex and striatum (by 40-67%), indicating reduced edema formation. Bumetanide also reduced infarct size, determined by TTC staining. These findings suggest that a luminal BBB Na-K-Cl cotransporter contributes to edema formation during cerebral ischemia.[3] |
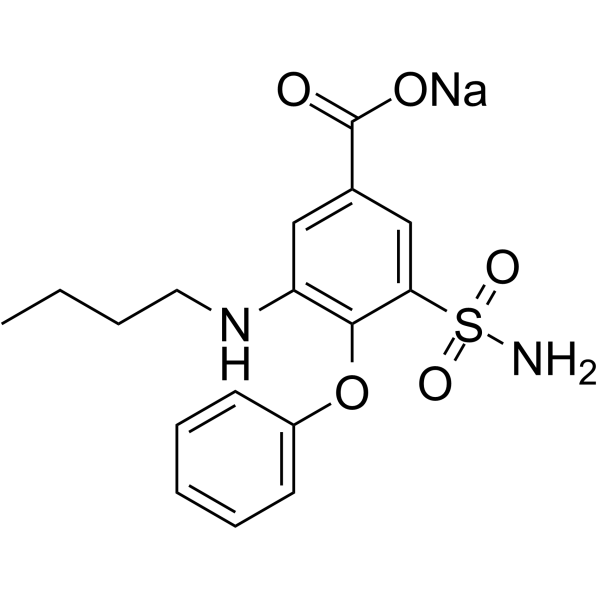
| 分子式 |
C17H19N2O5S-.NA+
|
|---|---|
| 分子量 |
386.39796
|
| 精确质量 |
386.091
|
| CAS号 |
28434-74-4
|
| 相关CAS号 |
Bumetanide;28395-03-1
|
| PubChem CID |
23696786
|
| 外观&性状 |
Typically exists as solid at room temperature
|
| 沸点 |
571.2ºC at 760mmHg
|
| 闪点 |
299.3ºC
|
| 蒸汽压 |
6.89E-14mmHg at 25°C
|
| LogP |
3.555
|
| tPSA |
129.93
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
7
|
| 可旋转键数目(RBC) |
8
|
| 重原子数目 |
26
|
| 分子复杂度/Complexity |
534
|
| 定义原子立体中心数目 |
0
|
| SMILES |
CCCCNC1=C(C(=CC(=C1)C(=O)O)S(=O)(=O)N)OC2=CC=CC=C2.[Na].[H]
|
| InChi Key |
QDFGOJHAQZEYQL-UHFFFAOYSA-M
|
| InChi Code |
InChI=1S/C17H20N2O5S.Na/c1-2-3-9-19-14-10-12(17(20)21)11-15(25(18,22)23)16(14)24-13-7-5-4-6-8-13;/h4-8,10-11,19H,2-3,9H2,1H3,(H,20,21)(H2,18,22,23);/q;+1/p-1
|
| 化学名 |
sodium;3-(butylamino)-4-phenoxy-5-sulfamoylbenzoate
|
| 别名 |
28434-74-4; Bumetanide sodium; Sodium 3-(aminosulphonyl)-5-(butylamino)-4-phenoxybenzoate; Bumetanide (sodium); 1QC8KM52D1; EINECS 249-015-6; UNII-1QC8KM52D1; SODIUM 3-(AMINOSULFONYL)-5-(BUTYLAMINO)-4-PHENOXYBENZOATE;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5880 mL | 12.9400 mL | 25.8799 mL | |
| 5 mM | 0.5176 mL | 2.5880 mL | 5.1760 mL | |
| 10 mM | 0.2588 mL | 1.2940 mL | 2.5880 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。