| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 2g |
|
||
| 5g |
|
||
| 10g |
|
||
| 25g |
|
||
| 50g |
|
||
| 100g |
|
||
| Other Sizes |
|

| 体外研究 (In Vitro) |
阿莫西林 (Amoxycillin)(1-100 µM;24 小时;嗜酸乳杆菌)以剂量依赖性方式减少活细胞并增加细胞壁破裂程度[1]。
|
|---|---|
| 体内研究 (In Vivo) |
当大鼠给予阿莫西林剂量为 7 毫克/公斤(ih;雌性 ICR/瑞士小鼠)时,大鼠存活率会增加,并且菌株数量受到抑制[2]。
给予阿莫西林(也称为阿莫西林)的瑞士白化小鼠阿莫西林)(1.6–9.5 mg/kg;口服;每天,持续 7 或 14 天)可预防沙眼衣原体感染[3]。 |
| 动物实验 |
Animal Model: Female ICR/Swiss mice[2]
Dosage: 7 mg/kg Administration: Subcutaneous injection: every eight hours for a full day Result: exhibited a dose-dependent inhibition on the number of bacteria. |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Amoxicillin is approximately 60% bioavailable. A 250mg dose of oral amoxicillin reaches a Cmax 3.93±1.13mg/L with a Tmax 1.31±0.33h and an AUC of 27.29±4.72mg\*h/L. A 875mg dose of oral amoxicillin reaches a Cmax 11.21±3.42mg/L with a Tmax 1.52±0.40h and an AUC of 55.04±12.68mg\*h/L. 125mg to 1g doses of amoxicillin are 70-78% eliminated in the urine after 6 hours. The central volume of distribution of amoxicillin is 27.7L. The mean clearance of amoxicillin is 21.3L/h. ... A 48 year-old woman was admitted because of pneumococcal meningitis. After 4 days on high-dose amoxicillin (320 mg/kg/day), she developed acute oliguric renal failure and amoxicillin crystallization was documented by infrared spectrometry. The outcome was favorable after amoxicillin dosage tapering, together with one single hemodialysis session and further hydratation. Amoxicillin is mainly excreted in the urine in its unchanged form. Amoxicillin diffuses readily into most body tissues and fluids, with the exception of brain and spinal fluid, except when meninges are inflamed. In blood serum, amoxicillin is approximately 20% protein-bound. Following a 1 gram dose and utilizing a special skin window technique to determine levels of the antibiotic, it was noted that therapeutic levels were found in the interstitial fluid. Although presence of food in the GI tract reportedly results in lower and delayed peak serum concentrations of amoxicillin, the total amount of drug absorbed does not appear to be affected. Amoxicillin was studied in normal subjects after intravenous, oral, and intramuscular administration of 250-, 500-, and 1,000-mg doses. Serum drug levels were analyzed using a two-compartment open model, as well as area under the curve (AUC) and urinary recovery. The variations of these pharmacokinetic parameters were then examined using the three-way analysis of variance and linear regression equations. These results confirmed nearly complete oral absorption: AUC was 93% of intravenous absorption, and urinary recovery was 86%. The intramuscular administration of amoxicillin results in complete and reliable absorption with peak drug levels, AUCs, and urinary recovery equivalent to oral dosage. The absorption of lyophilized amoxicillin after intramuscular injection resulted in an AUC that was 92% of intravenous absorption and urinary recovery of 91%. The peak serum levels, time to peak, and other pharmacokinetic parameters for intramuscular injection were nearly identical to those for oral administration. Kinetics of both intramuscular and oral administration exhibited dose-dependent absorption (absorption rate constant, 1.3/hr for 250 mg and 0.7/hr for 1,000 mg). This resulted in relatively later and lower peak serum levels for increasing dose. Total absorption, however, showed no dose dependence, as indicated by urinary recovery and AUC, which changed by less than 10%. For more Absorption, Distribution and Excretion (Complete) data for AMOXICILLIN (10 total), please visit the HSDB record page. Metabolism / Metabolites Incubation with human liver microsomes has lead to the detection of 7 metabolites. The M1 metabolite has undergone hydroxylation, M2 has undergone oxidative deamination, M3 to M5 have undergone oxidation of the aliphatic chain, M6 has undergone decarboxylation, and M7 has undergone glucuronidation. Biological Half-Life The half life of amoxicillin is 61.3 minutes. The half-life of amoxicillin is 61.3 minutes. |
| 毒性/毒理 (Toxicokinetics/TK) |
Hepatotoxicity
Rare instances of idiosyncratic liver injury have been reported in persons receiving the aminopenicillins including amoxicillin. Cases are characterized by a short latency period of a few days to as long as two weeks. The onset of liver injury can occur after the antibiotic is stopped. The serum enzyme pattern associated with aminopenicillin liver injury has included a hepatocellular pattern with marked elevations in ALT and AST, and minimal elevations in alkaline phosphatase and rapid recovery after withdrawal. In addition, cholestatic forms of hepatic injury with marked alkaline phosphatase elevations (as also seen with penicillin-induced liver injury) have also been described, some of which have been associated with prolonged cholestasis (Case 1). The onset of hepatic injury may be accompanied by signs or symptoms of hypersensitivity such as eosinophilia, rash and arthralgias, and in some cases is accompanied by toxic epidermal necrolysis or Stevens Johnson syndrome. Much more common than liver injury from amoxicillin alone is the typically cholestatic hepatitis that occurs after treatment with the combination of amoxicillin and clavulanate. Indeed, this combination is currently the most common cause of idiosyncratic acute liver injury in the United States, Europe and Australia. The injury, however, is usually attributed to the clavulanate rather than amoxicillin. The clinical features are similar but perhaps not completely the same. In cases of liver injury seeming due to amoxicillin, an extra effort should be made to make sure that it was not amoxicillin-clavulanate [Augmentin] that was taken. Likelihood score: B (highly likely but rare cause of clinically apparent liver injury). Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Limited information indicates that amoxicillin produces low levels in milk that are not expected to cause adverse effects in breastfed infants. Occasionally, rash and disruption of the infant's gastrointestinal flora, resulting in diarrhea or thrush, have been reported, but these effects have not been adequately evaluated. Amoxicillin is acceptable in nursing mothers. Amoxicillin powder for suspension reconstituted with breastmilk is absorbed as well as the powder reconstituted with water. ◉ Effects in Breastfed Infants In a telephone follow-up study, 25 nursing mothers reported taking amoxicillin (dosage unspecified). Three mothers reported diarrhea in their infants. No rashes or candidiasis were reported among the exposed infants. In contrast, a small, controlled, prospective study had mothers monitor their infants for signs of adverse effects (furring of the tongue, feeding difficulties, changes in stool frequency and consistency, diaper rash, and skin rash). Weight change and the development of jaundice were also recorded. No statistical differences in these parameters were found between the infants of the control mothers and those of mothers taking the related antibiotics, ampicillin or ampicillin-clavulanate. A prospective, controlled study asked mothers who called an information service about adverse reactions experienced by their breastfed infants. Of 40 infants exposed to amoxicillin in breastmilk, 2 developed diarrhea and 1 developed a rash. A study compared the breastfed infants of mothers taking amoxicillin to those taking a macrolide antibiotic. Adverse reactions occurred in 8.3% of the infants exposed to amoxicillin, which was similar to the rate in macrolide-exposed infants. Reactions included rash and somnolence. A 2-month-old infant breastfed since birth. His mother had taken many medications during pregnancy, but she did not recall their identity. She developed mastitis and was treated with amoxicillin/clavulanic acid 1 gram orally every 12 hours and gentamicin 160 mg intramuscularly once daily. The infant was breastfed for 10 minutes starting 15 minutes after the first dose of both drugs. About 20 minutes later, the infant developed a generalized urticaria which disappeared after 30 minutes. A few hours later, the infant breastfed again and the urticaria reappeared after 15 minutes and disappeared after an hour. After switching to formula feeding and no further infant exposure to penicillins, the reaction did not reappear with follow-up to 16 months of age. The adverse reaction was probably caused by the antibiotics in breastmilk. The drug that caused the reaction cannot be determined, but it was most likely the amoxicillin/clavulanic acid. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding Amoxicillin is 17% protein bound in serum. |
| 参考文献 | |
| 其他信息 |
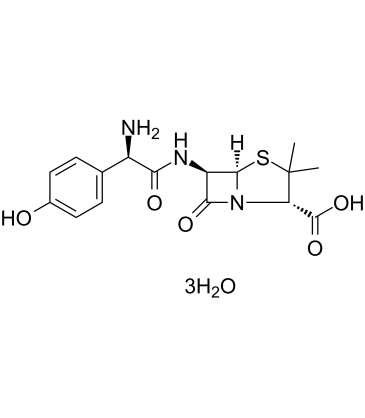
Amoxicillin is a penicillin in which the substituent at position 6 of the penam ring is a 2-amino-2-(4-hydroxyphenyl)acetamido group. It has a role as an antibacterial drug. It is a penicillin and a penicillin allergen. It is a conjugate acid of an amoxicillin(1-).
Amoxicillin is an antibacterial prescription medicine approved by the U.S. Food and Drug Administration (FDA) for the treatment of certain bacterial infections, such as community-acquired pneumonia; infections of the ear, nose and throat; infections of the genitourinary tract and infections of the skin and respiratory tract. Community-acquired pneumonia, a bacterial respiratory disease, can be an opportunistic infection (OI) of HIV. Amoxicillin, or BRL-2333, is a penicillin G derivative first described in the literature in 1972. Amoxicillin has similar activity to [penicillin] and [ampicillin], but leads to higher serum concentrations than ampicillin. Amoxicillin was granted FDA approval on 18 January 1974. Amoxicillin anhydrous is a Penicillin-class Antibacterial. Amoxicillin has been reported in Arundo donax and Apis cerana with data available. Amoxicillin is a broad-spectrum, semisynthetic aminopenicillin antibiotic with bactericidal activity. Amoxicillin binds to and inactivates penicillin-binding protein (PBP) 1A located on the inner membrane of the bacterial cell wall. Inactivation of PBPs interferes with the cross-linkage of peptidoglycan chains necessary for bacterial cell wall strength and rigidity. This interrupts bacterial cell wall synthesis and results in the weakening of the bacterial cell wall and causes cell lysis. Amoxicillin Anhydrous is the anhydrous form of a broad-spectrum, semisynthetic aminopenicillin antibiotic with bactericidal activity. Amoxicillin binds to and inactivates penicillin-binding proteins (PBPs) located on the inner membrane of the bacterial cell wall. Inactivation of PBPs interferes with the cross-linkage of peptidoglycan chains necessary for bacterial cell wall strength and rigidity. This interrupts bacterial cell wall synthesis and results in the weakening of the bacterial cell wall and causes cell lysis. A broad-spectrum semisynthetic antibiotic similar to AMPICILLIN except that its resistance to gastric acid permits higher serum levels with oral administration. Drug Indication Amoxicillin alone is indicated to treat susceptible bacterial infections of the ear, nose, throat, genitourinary tract, skin, skin structure, and lower respiratory tract. Amoxicillin is given with calvulanic acid to treat acute bacterial sinusitis, community acquired pneumonia, lower respiratory tract infections, acute bacterial otitis media, skin and skin structure infections, and urinary tract infections. Amoxicillin is given with omeprazole in the treatment of _Helicobacter pylori_ (_H. pylori_) infection. Amoxicillin is used in combination with [vonoprazan] and [clarithromycin] as co-packaged triple therapy or in combination with [vonoprazan] as co-packaged dual therapy to treat _H. pylori_ infection in adults. FDA Label Treatment of Helicobacter spp. infections Treatment of Helicobacter spp. infections Mechanism of Action Amoxicillin competitively inhibits penicillin-binding protein 1 and other high molecular weight penicillin binding proteins. Penicillin bind proteins are responsible for glycosyltransferase and transpeptidase reactions that lead to cross-linking of D-alanine and D-aspartic acid in bacterial cell walls. Without the action of penicillin binding proteins, bacteria upregulate autolytic enzymes and are unable to build and repair the cell wall, leading to bacteriocidal action. The penicillins and their metabolites are potent immunogens because of their ability to combine with proteins and act as haptens for acute antibody-mediated reactions. The most frequent (about 95 percent) or "major" determinant of penicillin allergy is the penicilloyl determinant produced by opening the beta-lactam ring of the penicillin. This allows linkage of the penicillin to protein at the amide group. "Minor" determinants (less frequent) are the other metabolites formed, including native penicillin and penicilloic acids. /Penicillins/ Amoxicillin is similar to penicillin in its bactericidal action against susceptible bacteria during the stage of active multiplication. It acts through the inhibition of cell wall biosynthesis that leads to the death of the bacteria. |
| 分子式 |
C16H25N3O8S
|
|---|---|
| 分子量 |
419.44
|
| 精确质量 |
419.136
|
| 元素分析 |
C, 45.82; H, 6.01; N, 10.02; O, 30.51; S, 7.64
|
| CAS号 |
61336-70-7
|
| 相关CAS号 |
Amoxicillin sodium;34642-77-8;Amoxicillin;26787-78-0;Amoxicillin-d4;2673270-36-3;Amoxicillin trihydrate mixture with potassium clavulanate (4:1);Amoxicillin arginine;59261-05-1
|
| PubChem CID |
33613
|
| 外观&性状 |
Crystals from water
|
| 密度 |
1.54g/cm3
|
| 沸点 |
743.2ºC at 760 mmHg Vapour
|
| 熔点 |
>200ºC (dec.)
|
| 闪点 |
403.3ºC
|
| 折射率 |
302 ° (C=0.1, H2O)
|
| LogP |
0.859
|
| tPSA |
185.95
|
| 氢键供体(HBD)数目 |
4
|
| 氢键受体(HBA)数目 |
7
|
| 可旋转键数目(RBC) |
4
|
| 重原子数目 |
25
|
| 分子复杂度/Complexity |
590
|
| 定义原子立体中心数目 |
4
|
| SMILES |
S1C(C([H])([H])[H])(C([H])([H])[H])[C@]([H])(C(=O)O[H])N2C([C@]([H])([C@@]12[H])N([H])C([C@@]([H])(C1C([H])=C([H])C(=C([H])C=1[H])O[H])N([H])[H])=O)=O.O([H])[H].O([H])[H].O([H])[H]
|
| InChi Key |
MQXQVCLAUDMCEF-CWLIKTDRSA-N
|
| InChi Code |
InChI=1S/C16H19N3O5S.3H2O/c1-16(2)11(15(23)24)19-13(22)10(14(19)25-16)18-12(21)9(17)7-3-5-8(20)6-4-7/h3-6,9-11,14,20H,17H2,1-2H3,(H,18,21)(H,23,24)3*1H2/t9-,10-,11+,14-/m1.../s1
|
| 化学名 |
(2S,5R,6R)-6-[[(2R)-2-Amino-2-(4-hydroxyphenyl)acetyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid trihydrate
|
| 别名 |
Larotid Amoxil Amoxipen Moxaline AmodexAmoxicillin Trihydrate Amoxicillin 3H2OAmoxicillin Trihydrate; Amoxicillin 3H2O; Larotid; Amoxil; Amoxipen; Moxaline; Amodex;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~10 mg/mL (~23.84 mM)
H2O : ~2 mg/mL (~4.77 mM) |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.08 mg/mL (4.96 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (4.96 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL 澄清 DMSO 储备液加入到 900 μL 玉米油中并混合均匀。 View More
配方 3 中的溶解度: ≥ 1 mg/mL (2.38 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: 10% DMSO+90% (20% SBE-β-CD in Saline): ≥ 2.08 mg/mL (4.96 mM) 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3841 mL | 11.9207 mL | 23.8413 mL | |
| 5 mM | 0.4768 mL | 2.3841 mL | 4.7683 mL | |
| 10 mM | 0.2384 mL | 1.1921 mL | 2.3841 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT05934227 | Enrolling by invitation | Drug: Amoxicillin 500 milligrams capsules plus metronidazole 400 milligrams Other: placebo |
Periodontal Pocket Periodontitis |
Federal University of Minas Gerais | 2023-07-01 | Not Applicable |
| NCT01933698 | Completed | Drug: amoxicillin | Bioequivalence of Amoxicillin | University of Campinas, Brazil | 2005-02 | Phase 4 |
| NCT01431989 | Completed | Drug: Amoxicillin powder for oral suspension (Clamoxyl®) 500mg/5mL Drug: Amoxil® 500mg/5mL powder for oral suspension |
Infections, Bacterial | GlaxoSmithKline | 2011-05-27 | Phase 1 |
| NCT05165212 | Withdrawn | Drug: Amoxicillin | Amoxicillin Allergy | State University of New York at Buffalo | 2024-12-31 | Phase 4 |
| NCT02853292 | Completed | Biological: urine collection | Amoxicillin Crystalluria | CHU de Reims | 2016-05 |
|
|
|