| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1mg |
|
||
| Other Sizes |
|
| 体外研究 (In Vitro) |
药物分子包括碳、氢和其他元素的稳定重同位素,主要作为药物开发过程中定量的示踪剂。由于其有可能改变药物的药代动力学和代谢特征,氘化引起了人们的关注[1]。
|
|---|---|
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
/MILK/ In goats, 1 hr after oral administration of 30 mg/kg bw nitrosodiethylamine, there were 11.4 mg/kg nitrosodiethylamine in milk and 11.9 mg/kg in blood. Only traces were found in milk and none in blood after 24 hr. Autoradiographic studies indicated that non-metabolized N-nitrosodiethylamine passed to fetuses with even distribution in most fetal tissues on all studied days of gestation (day 12, 14, 16, 16 and 18) in mice. Results also indicated metabolism of the substance in mucosa of fetal bronchial tree and liver on day 18 of gestation. Metabolism / Metabolites Inhibition of sulfotransferase by 2,6-dichloro-4-nitrophenol completely abolished the genotoxic potential of N-nitrosodiethanolamine in rat liver as indicated by the induction of DNA single-strand breaks. The DNA strand-breaking potential of N-nitroso-2-hydroxymorpholine, a metabolite of N-nitrosodiethanolamine formed by alcohol dehydrogenase -mediated oxidation, was also almost quantitatively abolished. In contrast to these beta-hydroxylated nitrosamines, the effectiveness of N-nitrosodiethylamine remained unaffected by 2,6-dichloro-4-nitrophenol with respect to its DNA damaging potential. ... A new activation mechanism for N-nitrosodiethanolamine is proposed: N-nitrosodiethanolamine is transformed at first by alcohol dehydrogenase into the cyclic hemiacetal N-nitroso-2-hydroxymorpholine. This cyclic beta-hydroxynitrosamine appears to be a substrate for sulfotransferase. The resulting sulfate conjugate is suggested to be ultimate genotoxic electrophile. However, the results do not exclude the possibility that N-nitrosodiethanolamine itself undergoes sulfate conjugation. Oxidative N-deethylation of NDEA accounts for the production of CO2 and alkylating species in vivo. The rate of metabolism of NDEA by slices of organs from rats and hamsters in vitro has been measured, and a correlation made between the degree of metabolism and the distribution of induced tumors. After administration of NDEA to rats or hamsters, several ethylated derivatives were produced in liver and kidney nucleic acids. These included 7-ethylguanine, O6-ethylguanine and 3-ethyladenine. ... Evidence suggests that nitrosodiethylamine requires metabolic activation in order to exert its carcinogenic and toxic effects. ... N-nitrosoethyl-N-(2-hydroxyethyl)amine and N-nitrosoethyl-N-(carboxymethyl)amine have been detected in urine of rats. ... Possible relationships between structure and metabolism of nitrosamines have been investigated in the rat small intestine. Isolated segments of jejunum and ileum were perfused from the luminal side for 2 hr with a Tyrode solution containing one of four symmetrical dialkylnitrosamines with 2-5 carbon atoms per side chain, all (14)C-labeled at the alpha position, or one of two unsymmetrical nitrosamines, N-nitroso-tert-butylmethylamine and N-nitrosomethylbenzylamine, (14)C-labeled in the methyl group. Besides measurement of (14)C to intestinal tissue, the absorbed fluid (absorbate) as well as the perfusion medium and tissue homogenates were analyzed by for the presence of polar metabolites to assess the intestinal metabolism of nitrosamines. Neither N-nitrosodiethylamine nor the two unsymmetrical nitrosamines were metabolized to any significant extent. Nitrosodiethylamine has known human metabolites that include N-Nitrosoethanamine. |
| 参考文献 | |
| 其他信息 |
n-Nitrosodiethylamine can cause cancer according to an independent committee of scientific and health experts.
N-nitrosodiethylamine is a clear slightly yellow liquid. Boiling point 175-177 °C. Can reasonably be anticipated to be a carcinogen. Used as a gasoline and lubricant additive and as an antioxidant and stabilizer in plastics. N-nitrosodiethylamine is a nitrosamine that is N-ethylethanamine substituted by a nitroso group at the N-atom. It has a role as a mutagen, a hepatotoxic agent and a carcinogenic agent. N-Nitrosodiethylamine is a synthetic light-sensitive, volatile, clear yellow oil that is soluble in water, lipids, and other organic solvents. It is used as gasoline and lubricant additive, antioxidant, and stabilizer for industry materials. When heated to decomposition, N-nitrosodiethylamine emits toxic fumes of nitrogen oxides. N-Nitrosodiethylamine affects DNA integrity, probably by alkylation, and is used in experimental research to induce liver tumorigenesis. It is considered to be reasonably anticipated to be a human carcinogen. (NCI05) A nitrosamine derivative with alkylating, carcinogenic, and mutagenic properties. Mechanism of Action ... It is shown that the two nitrosamines N-nitrosodiethylamine and 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone bind to nicotinic cholinergic receptors in hamster lung. Binding of the nitrosamines as well as nicotine to this receptor stimulates proliferation of human lung carcinoid cells in vitro. These data suggest chronic stimulation of nicotinic receptors by nicotine and nitrosamines in smokers as one of the molecular events responsible for stimulation of neuroendocrine cell proliferation and ultimately the development of lung tumors with neuroendocrine differentiation. ... |
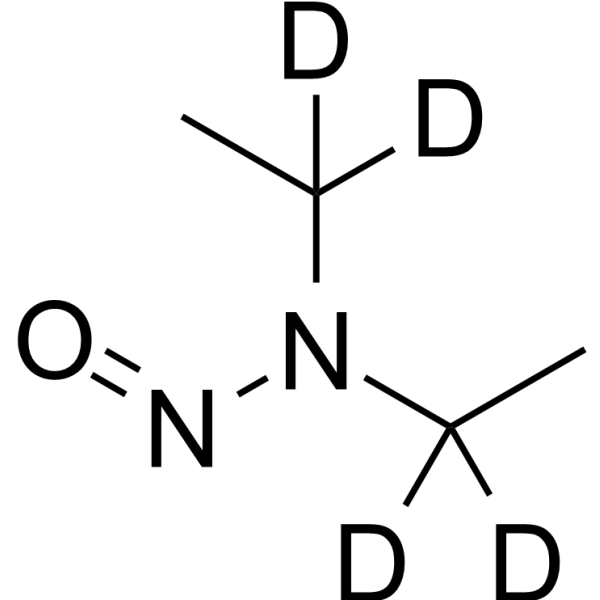
| 分子式 |
C4H6D4N2O
|
|---|---|
| 分子量 |
106.16
|
| 精确质量 |
106.104
|
| CAS号 |
1346603-41-5
|
| 相关CAS号 |
N-Nitrosodiethylamine;55-18-5
|
| PubChem CID |
5921
|
| 外观&性状 |
Yellow oil
Slightly yellow liquid |
| 密度 |
0.9±0.1 g/cm3
|
| 沸点 |
173.9±9.0 °C at 760 mmHg
|
| 闪点 |
59.0±18.7 °C
|
| 蒸汽压 |
1.7±0.3 mmHg at 25°C
|
| 折射率 |
1.442
|
| LogP |
0.42
|
| tPSA |
32.7
|
| 氢键供体(HBD)数目 |
0
|
| 氢键受体(HBA)数目 |
3
|
| 可旋转键数目(RBC) |
2
|
| 重原子数目 |
7
|
| 分子复杂度/Complexity |
51.7
|
| 定义原子立体中心数目 |
0
|
| SMILES |
CCN(CC)N=O
|
| InChi Key |
WBNQDOYYEUMPFS-VEPVEJTGSA-N
|
| InChi Code |
InChI=1S/C4H10N2O/c1-3-6(4-2)5-7/h3-4H2,1-2H3/i1D2,3D2
|
| 化学名 |
N-ethyl-N-(1,1,2,2-tetradeuterioethyl)nitrous amide
|
| 别名 |
DEN-d4; Diethylnitrosamine-d4; N-Nitrosodiethylamine-d4
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 9.4197 mL | 47.0987 mL | 94.1974 mL | |
| 5 mM | 1.8839 mL | 9.4197 mL | 18.8395 mL | |
| 10 mM | 0.9420 mL | 4.7099 mL | 9.4197 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。